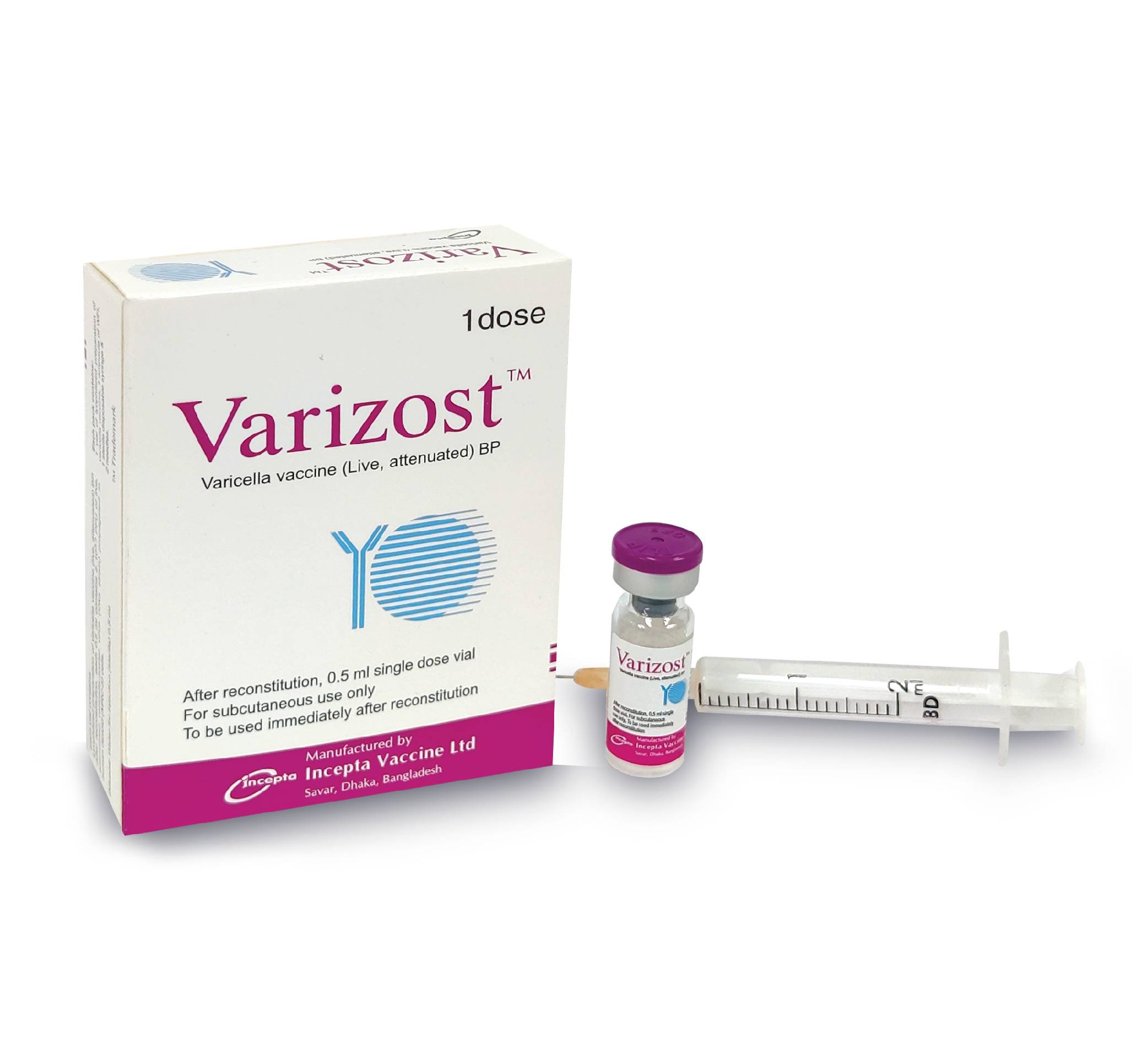
Presentation:
VarizostTM : Each vial contains lyophilized preparation of varicella vaccine (live, attenuated) BP. After reconstitution, each 0.5 ml contains ≥ 103.3 PFU of live, attenuated varicella-zoster virus (Oka strain) propagated in human diploid (MRC-5) cells.
Description:
Varizost is a lyophilized preparation (with appropriate stabilizer) of the live, attenuated varicella-zoster virus (Oka strain) obtained by propagation of the virus in human diploid (MRC-5) cell culture. After reconstitution, the solution should be homogenous, clear solution and free from any foreign particulate matter.
Indications:
The vaccine is indicated for the active immunization against varicella of adults and children from 12 months of age. Varicella vaccine may also be used for the active immunization against varicella of susceptible high risk patients and their susceptible healthy close contacts.
Dosage and Administration:
One immunizing dose contains 0.5 ml of reconstituted vaccine. The reconstituted vaccine should be used subcutaneously for both children and adults. Do not inject the vaccine intramuscularly or intravenously.
Children from 12 months of age, adolescents & adults: 2 doses (0.5 ml each) of varicella vaccine to ensure optimal protection against varicella. It is preferable to administer the second dose at least 6 weeks after the first dose but in no circumstances less than 4 weeks.
Side Effects:
Local and systemic reactions:
Very common: fever which happens in 1-2 weeks after inoculation, mostly mild, can be relieved automatically after lasting 1-2 days without any treatment, patients need rest if necessary and more water, keeping warm in case of secondary infection; for moderate fever or fever time more than 48 hours, it can be treated by physical method or medicine.
Common: headache, weakness, reddened on injection site, pain, swelling, pruritus.
Occasional: allergic reaction, rash, induration, haphalgesia. Transient rash happens on a few of children after 6-12 days of inoculation which can be relieved automatically within 2 days without any special treatment, it can carry out symptomatic treatment if necessary.
Precautions:
- It is advisable to have a solution of epinephrine available in case of rare anaphylactic reaction.
- Generally speaking, it is advisable to keep the subject under medical supervision for 30 minutes following vaccination of this product.
- Transmission of vaccinal virus only occurs in extremely rare cases. All patients who may develop varicella, especially the patients suffering from cutaneous reaction two to three weeks after vaccination, should avoid contacting with patients suffering from leukemia or who are undergoing immune-suppressant therapy, or pregnant women especially in the first three months of pregnancy.
- Administrated subcutaneously, not intradermally and never, under any circumstances, intravenously.
- Avoid any disinfectant to contact the vaccine of this product during opening the vaccine vials and carrying the injection. Alcohol and any other disinfectants may inactivate the attenuated virus; thus the vaccination should be applied right after ensuring the complete evaporation of the disinfectant away from skin.
- Not allow to administrate injection in conditions of incomplete dissolution of this product, foreign matters observed in the dissolved vaccine, cracked glass vial and unclear label of glass vial.
- Women of child-bearing age can be vaccinated only if appropriately contraceptive measures have been taken for at least 3 months following vaccination.
- Vaccine of this product should be administered immediately when opening the vaccine vials; in special circumstances, the vaccine can be placed at 2-8 0C, and should be used within 30 minutes, the remnant vaccine should be discarded.
- Administration of other live attenuated vaccine should keep at least one-month interval after vaccination with this vaccine of product; however, this vaccine of product can be administered simultaneously with live attenuated vaccines of measles, rubella and mumps.
- Patients suffering from leucocythemia, tumor or immunodeficiency should restrainedly use under doctors’ guidance.
- Patients in the following conditions should restrainedly use: person or the person’s family has convulsions history, patients suffering from chronic disease, history of epilepsy, patients with allergic constitution, women in lactation.
- Patients injected with immunoglobulin should administer this vaccine of product at least after 3-month interval to avoid influencing the immunization effects.
Freezing is forbidden.
- Syncope (fainting) can occur following, or even before, any vaccination especially in adolescents as a psychogenic response to the needle injection. This can be accompanied by several neurological signs such as transient visual disturbance, paraesthesia and tonic-clonic limb movements during recovery.
- Serologic studies indicate that, the vaccine does not completely protect all individuals from naturally-acquired varicella and cannot be expected to provide maximal protection against infection with varicella zoster virus until about six weeks after the second dose.
- The Oka vaccine viral strain has recently been shown to be sensitive to acyclovir. Vaccination may be considered in patients with selected immune deficiencies where the benefits outweigh the risks.
Contraindications:
- Subjects with known hypersensitivity to any constituent of this product including neomycin.
- Women during pregnancy.
- Subjects suffering from serious diseases (acute or chronic infection), fever and any advanced immune disease.
- Subjects treated with steroidal drug.
- Subjects with a total lymphocyte count of less than 1200 per cubic meter or presenting other signs of cellular immunodeficiency.
- Subjects with known history of congenital immune disease or having closely touched with the family member who has a history of this disease.
- The effects of this product will be cut down for use of whole blood, plasma or immunoglobulin within 5 months before vaccination or within 3 weeks after vaccination.
- Avoid the use of salicylate within 6 weeks following vaccination of this product.
- Subjects suffering from cerebropathy and uncontrolled epilepsy and other progressive neurological diseases.
Use in Pregnancy and Lactation:
Pregnancy: Pregnant women must not be vaccinated with varicella vaccine. Pregnancy should be avoided for three months after vaccination.
Lactation: There are no data regarding the use in breast-feeding mother.
Over Dosage:
Not applicable
Storage:
- Keep out of the reach and sight of children
Store and transport at +2°C to +8°C. Do not freeze
- Protect from light
Commercial Pack:
VarizostTM : Each box contains 1 vial of lyophilized preparation of varicella vaccine, 1 ampoule containing 0.5 ml WFI, a sterile disposable syringe & 2 needles.
Others:
- The vaccine is for subcutaneous use only and must not be administered intravenously or intramuscularly
- To reconstitute the vaccine, transfer the supplied diluent in the vial containing the freeze-dried preparation
- The vaccine should be reconstituted only with the supplied diluent Do not shake vigorously
- The reconstituted vaccine should be inspected for any foreign particulate matter prior to administration



.png)